|
 |
|
|
 |
||
| Annual Report | ||
 |
News | |
| MDRC Administration | ||
 |
Directory / Bottin | |
 |
Contact Us | |
 |
Directions / Maps | |
 |
Steering Committee | |
| Research | ||
 |
Events & Seminars | |
 |
MDRC at the forefront | |
 |
McGarry Lecture | |
 |
Cahill Lecture | |
 |
MDRC Core Facilities | |
 |
Scientific Links | |
 |
Education & Employment | |
| Funding | ||
 |
MDRC Grants | |
 |
Donations | |
 |
Non-Profit & Industrial Partners | |
| Medical Information | ||
 |
JDRF | |
 |
CDA | |
 |
Diabetes Quebec | |
 |
ADA | |
 |
IDF | |
 |
NIH / NIDDK | |
 |
Alfediam | |
 |
EASD | |
| Home / About Us > Dr Jennifer Estall | 
Contact info Dr Jennifer Estall Tel: 1-514-987-5688
Research keywords
|
|
Jennifer Estall, PhD
Assistant Professor of Medicine
Biographical Sketch
After completing her undergraduate degree in Pharmacology at the University of Toronto in 2000, Dr Estall joined the lab of Dr Daniel Drucker within the Banting and Best Diabetes Centre, University of Toronto. She completed her PhD in 2005, with focus on GPCR structure and hormone receptor desensitization. Dr Estall moved to Boston in 2006 to join the laboratory of Dr Bruce Spiegelman at the Dana-Farber Cancer Institute/ Harvard Medical School as a post-doctoral fellow, where she investigated how disruption of transcriptional co-regulator activity contributes to the development of obesity and diabetes. She started her own lab at the Institut de recherches clinique de Montr�al (IRCM) as Assistant Research Professor in 2011, where she is interested in mitochondrial biology and the transcriptional regulation of metabolism. Her primary academic affiliation is with the Universit� de Montr�al, Faculty of Medicine and she holds an adjunct appointment at McGill University within the Division of Experimental Medicine. Selected Scientific Contributions
Dr Estall is particularly interested in how changing activity of peroxisome proliferator-activated receptor-gamma co-activators (PGC-1α and PGC-1β) within metabolically active tissues (i.e. liver, muscle, β-cells) impact whole body glucose homeostasis. Through generation and detailed phenotypic analysis of mice with tissue-specific reduction of PGC-1s, she uncovered a pathway by which reduced co-activator activity in liver impacts both hepatic and peripheral insulin sensitivity. Furthermore, she discovered that PGC-1α negatively influences gene expression in the liver through regulated production of heme, a ligand for the transcriptional repressor Rev-Erbα. These discoveries, in addition to expanding our understanding of how disrupted transcriptional signaling contributes to metabolic syndrome, also have implications for the role of PGCs in regulating the circadian clock and targeting of hepatic PGC-1α to treat diabetes. In addition to her own projects, Dr Estall worked closely with other research groups to expand our knowledge of the molecular events that underlie the development of diabetes and identify new treatment options. These collaborative efforts have helped to determine how changes in the browning capacity of white adipose tissue impacts insulin sensitivity and identified a novel class of PPARγ agonists with great therapeutic potential for type 2 diabetes. Research Interests
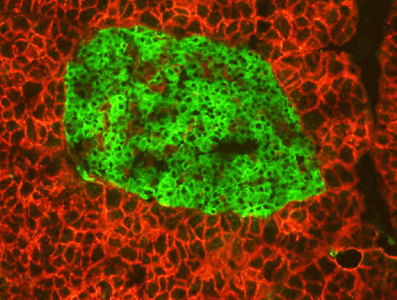
Both Type 1 and Type 2 diabetes involve abnormalities in energy homeostasis occurring at the cellular level. In particular, the uncontrolled production of insulin, destruction of β-cells, ectopic lipid accumulation, and insulin resistance in metabolically active tissues are central mechanisms in the pathogenesis of this disease. In addition, metabolic dysfunction in organs such as muscle and liver also play a significant role in disease progression and can directly impact β-cell health and function. The consequences of diabetes on overall health are well characterized; however, the initiating signals and steps within affected cells leading to pathology remain poorly understood at the molecular level. Consequently, biomarkers that reliably detect early islet dysfunction do not currently exist, limiting our ability to predict disease development prior to symptom appearance. Diabetes is caused by abnormalities in energy homeostasis at the cellular level. Using genetic models, biochemistry, proteomics, and gene expression pathway analysis, our lab focuses on identifying key molecules implicated in the onset of diabetes and other metabolic diseases. We focus our efforts toward understanding how deregulated metabolism contributes to the destruction of insulin-producing β-cells in the pancreas and fatty liver disease, two pathologies that play major roles in the severity of diabetes. We have a particular interest in how environment and genetics work together to either protect against or worsen disease. Our goals are to: 1. Better understand the molecular changes that occur within diseased tissues of diabetics; 2. Identify new molecular factors and genetic programs important for islet and liver function; 3. Reveal new therapeutic targets in an effort to prevent or lessen disease progression.
|
|
| � Montreal Diabetes Research Center 2018 |
| Home / About Us News Directory / Bottin Contact Us |